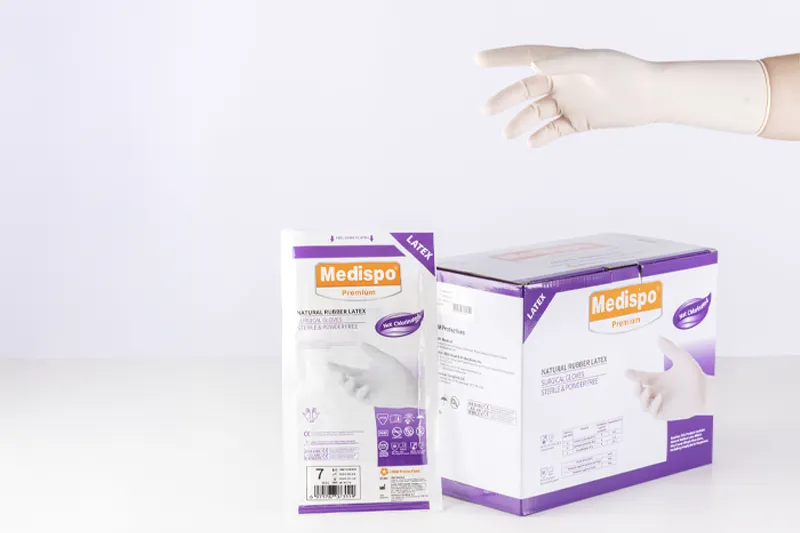
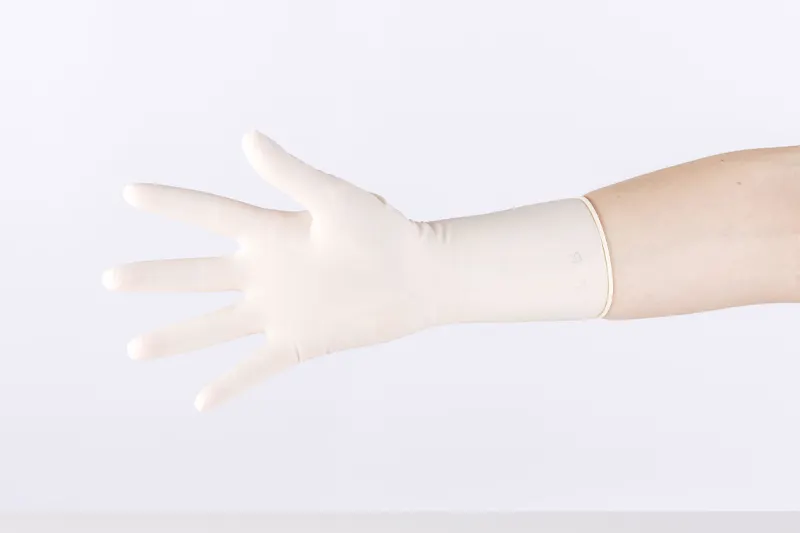
Sterile, powder-free latex surgical gloves, MEDISPO PREMIUM brand.
Dual-use product: Class IIa Medical Device and Category III Personal Protective Equipment.
Uses and applications:
For protection against biological and chemical risks during surgical procedures, invasive procedures, or open wound examinations. Suitable for use in contact with chemotherapy drugs (cytostatics). It is also recommended for use in any environment or sector requiring sterility or higher levels of hygiene, such as laboratories, cleanrooms, or veterinary clinics.
Regulatory Compliance:
It complies with European Regulations 745/2017 (MD), 425/2016 (PPE) and European Directive 93/42/EC, as well as with applicable standards EN ISO 374 (1-2-4-5), EN ISO 16523-1, EN 455 (1-2-3-4), EN ISO 21420, ASTM D 6978-05, among others.
Powder free
Sectors: