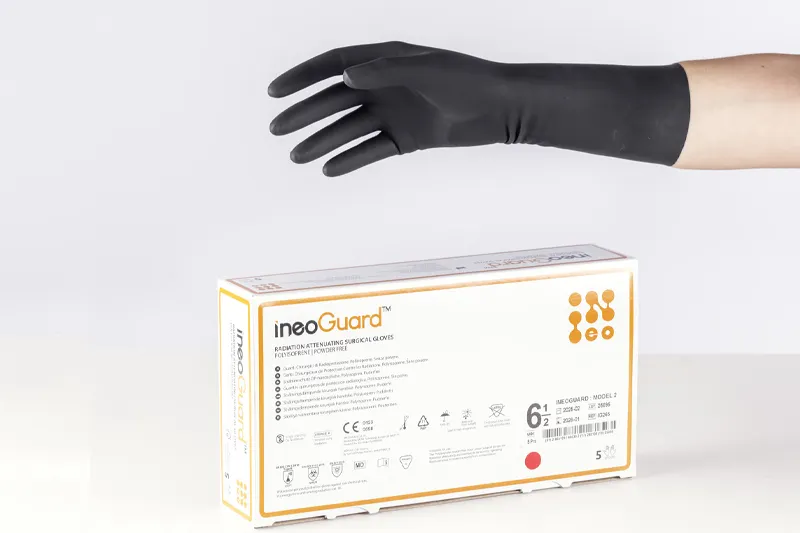
The development of less invasive approaches in the operating room is leading to more fluorescence-guided procedures. Surgical team members are exposed to scattering ionizing radiation and, occasionally, primary beams, with the hands being one of the most affected areas. IneoGuard is a sterile, latex-free, and powder-free surgical glove with an advanced polyisoprene formulation containing tungsten particles. This provides enhanced radiation attenuation, reducing the operator's exposure to harmful scattering or secondary ionizing radiation during fluoroscopic procedures.
Furthermore, its unique material composition offers greater flexibility, allowing for excellent tactile sensitivity and prolonged use without hand fatigue. The micro-textured fingertips provide optimal grip control. Two models are available, depending on thickness.
Black, sterile, powder-free, synthetic polyisoprene surgical glove, IneoGuard brand.
Free of natural latex and other additives such as rubber vulcanization derivatives that can cause allergies or irritations, such as Mercaptobenzothiazoles (MBT) and Guanidines, which cause Type I and IV allergies. Offers protection against biological and chemical risks (Type B).
Regulatory Compliance :
Dual-use product: Class IIa Medical Device and Category III Personal Protective Equipment.
Complies with European Regulations 745/2017 (MD) and 425/2016 (PPE), as well as applicable standards EN ISO 374 (1-2-4-5), EN ISO 16523-1, EN 455 (1-2-3-4), and EN ISO 21420, among others.
Powder free
Sectores: